 
The polarity of the water molecule and its resulting hydrogen bonding make water a unique substance with special properties that are intimately tied to the processes of life. These peculiar effects are due to the highly directional bonding of water molecules via the hydrogen bonds: ice and liquid water at low temperature have comparatively low-density, low-energy open lattice structures. Without it, life as we know it simply would not exist. Regular, hexagonal ice is also less dense than liquid waterupon freezing, the density of water decreases by about 9. Approximately 60–70 percent of the human body is made up of water. Water is one of the more abundant molecules and the one most critical to life on Earth. Salinity is a natural factor of impressive. 
Evaporation builds saltiness and density whereas the expansion of freshwater diminishes saltiness and density. Note: We could say that saltiness can diminish from the dissolving of polar ice or increase from the freezing of polar ice. Why do scientists spend time looking for water on other planets? Why is water so important? It is because water is essential to life as we know it. As salinity increases, the water density could also increase. Discuss the role of acids, bases, and buffers in homeostasis.Provide examples of water’s cohesive and adhesive properties.If you want to know the buoyant force on an object, you only need to determine the weight of the fluid displaced by the object. 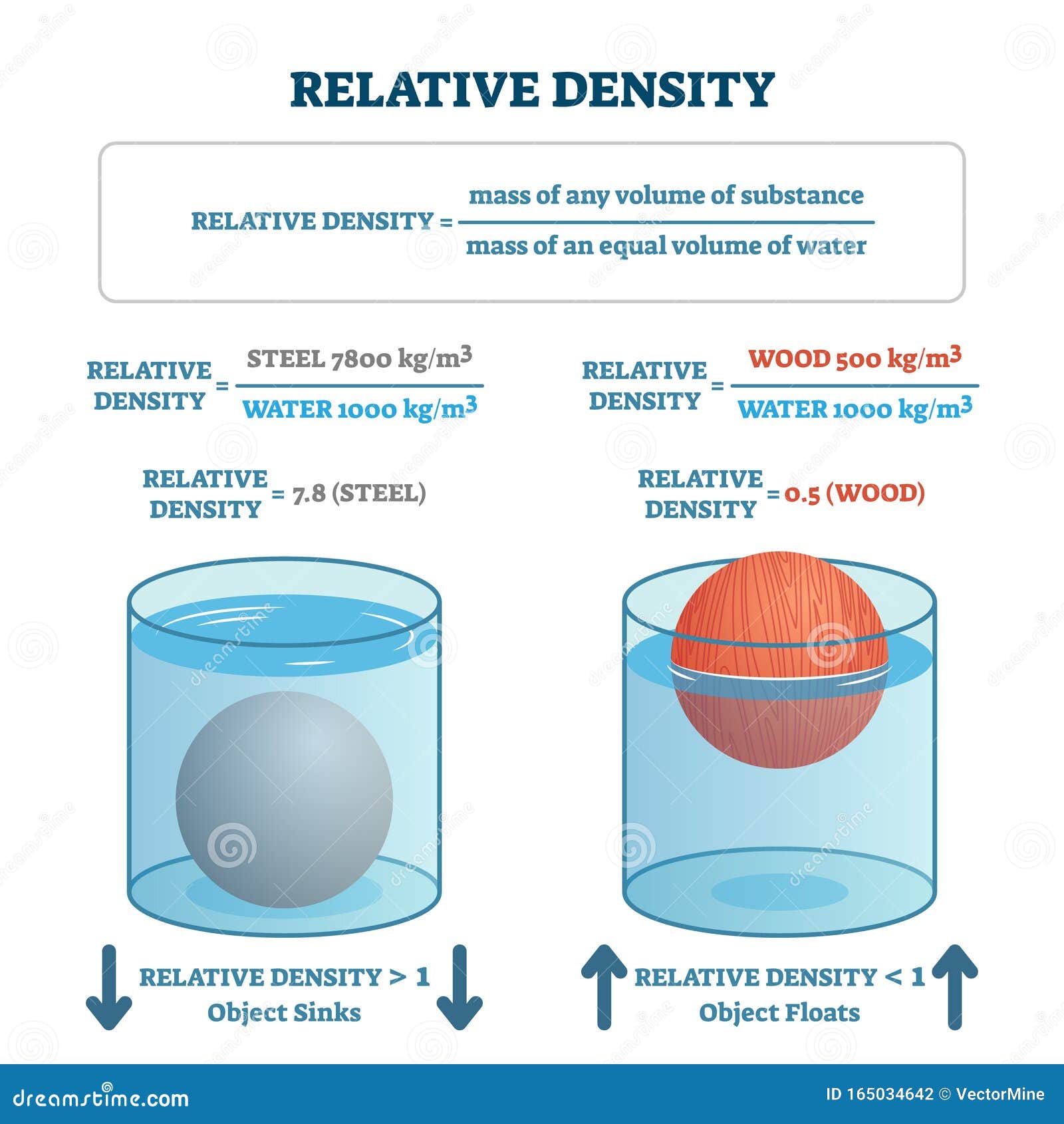
The simplicity and power of this idea is striking.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
